

Bio and Brain Engineering
mRNA vaccine & therapeutics, drug delivery, biomaterials, immunoengineeringTeacher start-up company : Exopert (https://www.exopert.com), De novo Biotherapeutics (http://www.denovobio.co.kr/en/index.php)
Research contents
Research results
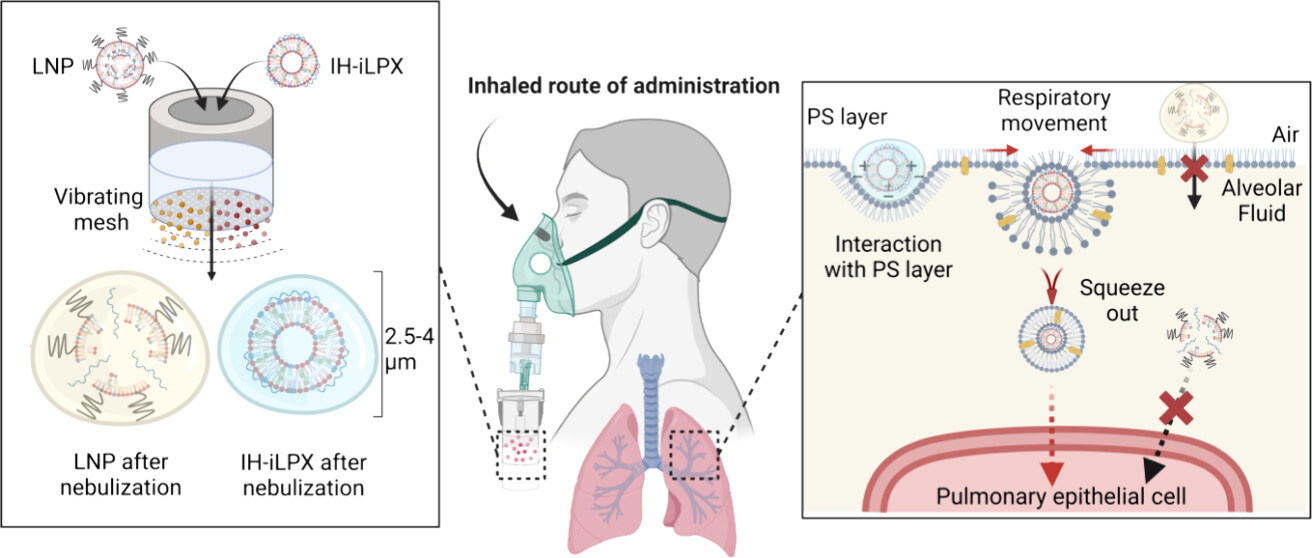
The delivery of mRNA into the lungs is the key to solving infectious and intractable diseases that frequently occur in the lungs. Since inhalation using a nebulizer is the most promising method for mRNA delivery into the lungs, there have been many attempts toward adapting lipid nanoparticles for mRNA inhalation. However, conventional lipid nanoparticles, which have shown great effectiveness for systemic delivery of mRNA and intramuscular vaccination, are not effective for pulmonary delivery due to their structural instability during nebulization and their inability to adapt to the pulmonary microenvironment. To address these issues, we developed an ionizable liposome-mRNA lipocomplex (iLPX). iLPX has a highly ordered lipid bilayer structure, which increases stability during nebulization, and its poly(ethylene glycol)-free composition allows it to infiltrate the low serum environment and the pulmonary surfactant layer in the lungs. We selected an inhalation-optimized iLPX using a multistep screening procedure that mimics the pulmonary delivery process of inhaled nanoparticles. The IH- iLPX showed a higher transfection efficiency in the lungs compared to conventional lipid nanoparticles after inhalation with no observed toxicity in vivo. Furthermore, analysis of lung distribution revealed even protein expression in the deep lungs, with effective delivery to epithelial cells. This study provides insights into the challenges and solutions related to the development of inhaled mRNA pulmonary therapeutics.
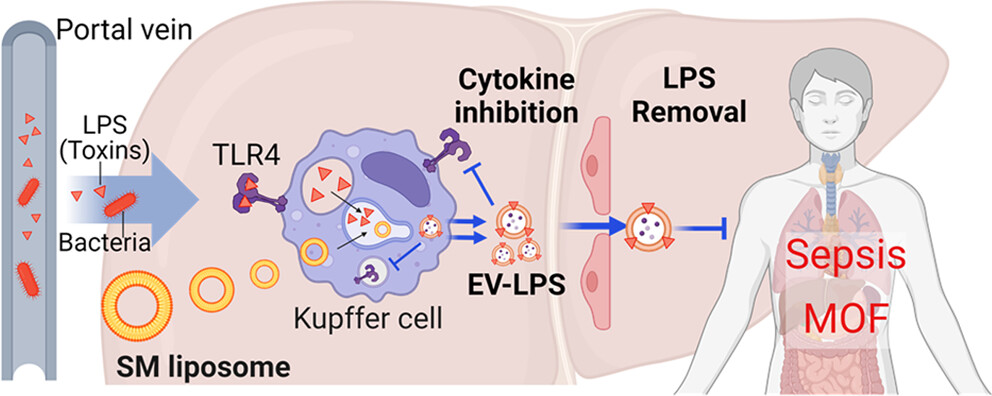
Sepsis remains a leading cause of death worldwide, and treatment options are limited. Antivirulence strategies that target toxins have emerged as a promising next-generation therapeutic approach due to the potential shown in preclinical studies. Here, we present the efficacy of sphingomyelin (SM) liposomes in modulating inflammatory responses and inhibiting disease progression in lipopolysaccharide (LPS)-induced sepsis. Our findings demonstrate that SM liposomes promote the exocytosis of LPS from LPS-challenged macrophages by enhancing the secretion of extracellular vesicles (EVs). SM liposomes further reduce the production of pro-inflammatory cytokines in LPS-challenged macrophages through EV-mediated LPS exocytosis. In an LPS-induced sepsis mouse model, we show that the systemic administration of SM liposomes significantly reduces local and systemic inflammation, improving the survival rate of mice. Our results illustrate the potential of SM liposomes as an antivirulence agent for the treatment of bacterial infections and sepsis.
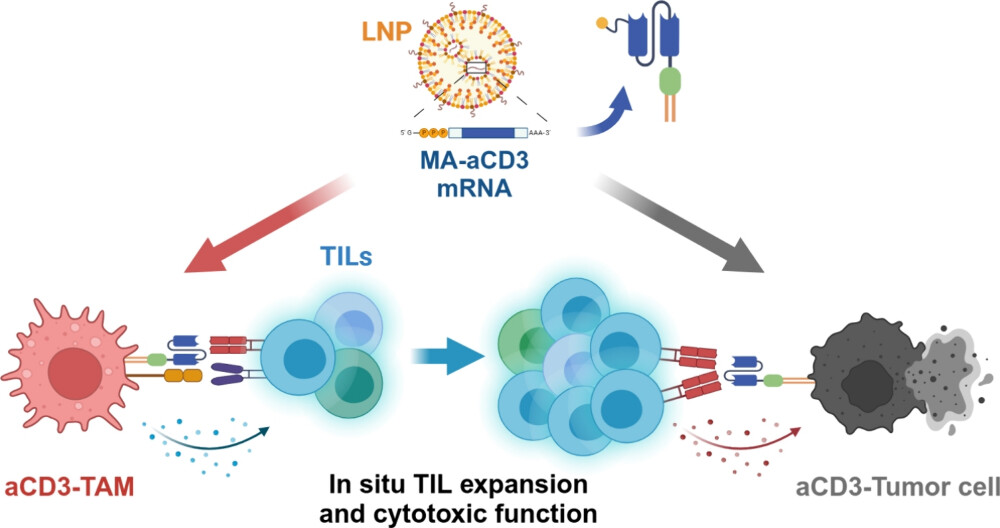
Tumor-infiltrating lymphocyte (TIL) therapy has shown promising responses in clinical trials for highly aggressive cancers such as advanced melanoma and metastatic colorectal cancer. However, TIL therapy is still limited in clinical practice due to the complex ex vivo cell preparation process. Here, we report an "in situ TIL therapy" for the treatment of solid tumors. We utilized lipid nanoparticles for the delivery of an mRNA encoding membrane-anchored anti-CD3 single-chain variable fragment (scFv) (MA-aCD3), efficiently engineering both tumor-associated macrophages (TAMs) and tumor cells following intratumoral delivery. Expression of MA-aCD3 resulted in enhanced TIL activation, proliferation, and tumor cell engagement directly within the tumor microenvironment. In B16F10 and MC38 tumor models, concurrent expression of MA-aCD3 on TAMs and tumor cells mediated by mRNA delivery resulted in significant antitumor effects via in situ polyclonal CD8+ TIL expansion and directed cytotoxic effector functions. In addition, combinatorial treatment of MA-aCD3-encoding mRNA and antiprogrammed cell death 1 (anti-PD-1) antibodies exhibited synergistic antitumor effects on anti-PD-1 refractory B16F10 tumors. Together, our findings suggest that in situ TIL therapy is a practical and effective mRNA-based therapeutic modality for the treatment of solid tumors
