

GSMSE(Graduate School of Medical Science & Engineering)
Somatic Mosaicisim in Intractable Neurological Disorders & RNA therapeuticsTeacher start-up company : Sovargen (http://www.sovargen.com/)
Research contents
Research results

- Composition for preventing or treating intractable epilepsy comprising mTOR inhibitor (Korea, 10-2343918)
- Animal model of brain tumor and manufacturing method of animal model (Korea, 10-2138131)
- Composition for Diagnosis, Prevention, or Treatment of Ganglioglioma and Disease Related Thereto (Japan,6941688)
- Composition for the diagnosis of ganglioglioma and related diseases (Korea, 10-1987202)
- Ganglioglioma-induced animal model and drug screening using the same (Korea, 10-1987203)
- Brain somatic mutations associated to epilepsy and uses thereof (Korea, 10-1771168)
- Prediction Method for using tissue origin of gliblastoma detecting genitic mutation animal model for gliblastoma(Korea 10-1838394)
- Compasition for prevention or treatment of intractable epilepsy comprising MTOR inhobotor(France,EU,UK,Germany –3266455)
- Disease model of intractable epilepsy(USA, 9629346)
- Animal model for epilepsy and method for producing the same (Korea, 10-1678961)
- Brain somatic mutations associated to epilepsy and uses thereof (Korea, 10-1739104)
- Biomaker for diagnosing epilepsy (Korea, 10-1547307)
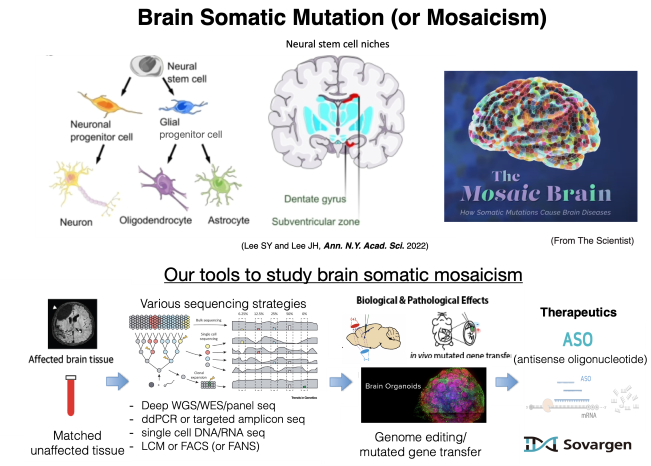
Our lab has made significant contribution to the field of ‘brain somatic mutations (or mosaicism) in focal epilepsy’, leading to a paradigm shift in understanding genetic etiology of intractable focal epilepsy. Our pioneering works provided the first direct evidence that low-level of somatic mutations in MTOR pathway genes arise from neural stem cells during development in the affected focal cortical regions and cause hemimegalencephaly (HME) and focal cortical dysplasia type II (FCDII) with intractable focal epilepsy (Lee et al, Nat Genet 2012; Lim et al, Nat Med 2015; Lim et al, Am J Hum Genet 2017). This condition presenting dysmorphic neurons and cortical dyslamination in the brain pathology is the most common cause of childhood intractable epilepsy subjected to surgical treatment. our lab also developed the new diagnostic tools to accurately detect extremely low-level somatic mutations, sufficient to cause intractable focal epilepsy (Kim et al, Nat Commun 2019; Sim et al, Acta Neuropathol 2019; Kim et al, Annal Neurol 2023). Furthermore, our lab recently determined the minimum threshold of somatic mosaicism leading to focal epilepsy, which can be less than 0.5% of mutation burden in patient’s brain tissues (Kim et al., Brain 2024), allowing us to identify the genetic causes in up to 80% of FCD type II patients (Kim et al, Annal Neurol 2023).Reflecting our pioneering works and others, the ILAE (International League Against Epilepsy) task force on FCD classification, where I served as a committee member in charge of the genetic part, recently updated the new classification and diagnostic criteria by adding the genetic finding of somatic mutations in FCD (Najm et al, Epilepsia 2022). To provide minimally invasive tools for genetic diagnosis, our lab demonstrated the possibility of detecting low-level somatic mutations in patient’s cerebrospinal fluid (CSF) in addition to surgical brain tissues (Kim et al, Annal Neurol 2021). Beside the identification of genetic causes, his lab identified mTOR as a promising therapeutic target for intractable epilepsy in FCDII using various mouse models of somatic mosaicism in mTOR pathway genes (Lim et al, Nat Med 2015; Lim et al, Am J Hum Genet 2017). Sovargen, a KAIST faculty start-up that Prof. Lee cofounded, is currently developing ASO therapy targeting mTOR in FCDII. Taken together, his seminal studies of somatic mutations in HME and FCDII lay the foundation for understanding somatic mosaicism as a new genetic cause of epilepsy.
The concept of brain somatic mosaicism in neurological disorders extends beyond FCD with intractable focal epilepsy. He expanded the idea of brain somatic mosaicism arising from neural stem cells to include low-grade epilepsy associated tumors (LEAT), glioblastoma (GBM), schizophrenia (SCZ), autism, and Alzheimer’s Diseases (AD). Particularly, GBM is the most devastating and incurable brain tumor, with a median overall survival of fifteen months. Identifying the cell of origin that harbors mutations that drive GBM could provide a fundamental basis for understanding disease progression and developing new treatments. Regarding this, his lab showed astrocyte-like neural stem cells in the subventricular zone to be the cell of origin that contains the driver mutations of human GBM (Lee et al, Nature 2018). These studies shows that his discovery of somatic mosaicism in intractable focal epilepsy indeed provides new insight into the molecular genetic pathogenesis of other neurological disorders and emphasizes the importance of basic science research in epilepsy, which can have broad impacts on other fields of neurological disorders
